
Retatrutide in Albuquerque, New Mexico
Retatrutide: A Next-Generation Triple-Action Weight Loss Medication
What Patients Should Know Before It Reaches the Clinic
Retatrutide is one of the most promising medications being studied for medical weight loss, blood sugar control, fatty liver, and metabolic health. It is often described as a triple-agonist medication because it activates three hormone receptor pathways:
GLP-1 to help reduce appetite, slow stomach emptying, improve satiety, and support blood sugar regulation.
GIP to support insulin signaling and metabolic regulation.
Glucagon to increase energy expenditure and potentially improve fat metabolism, especially in the liver.
This makes Retatrutide different from Semaglutide and Tirzepatide. Semaglutide works primarily through GLP-1. Tirzepatide works through GLP-1 and GIP. Retatrutide adds glucagon receptor activity, which may explain why early studies are showing such powerful effects on weight loss and metabolic health.
However, Retatrutide is still investigational. It is not currently FDA approved and is not legally available for routine prescribing outside of clinical trials. Eli Lilly states that Retatrutide is being evaluated in clinical trials and is not yet approved by the FDA
Benefits Being Studied With Retatrutide
Substantial Weight Loss
In a Phase 2 obesity trial published in The New England Journal of Medicine, Retatrutide produced major weight loss over 48 weeks. At the highest studied dose, participants lost up to 24.2% of body weight at 48 weeks.
In Lilly’s first successful Phase 3 obesity-related trial, TRIUMPH-4, adults with obesity or overweight and knee osteoarthritis taking Retatrutide 12 mg lost an average of 28.7% of body weight at 68 weeks.
These results are impressive, but they do not mean Retatrutide is right for everyone. Stronger weight loss can also require more careful medical monitoring, nutritional guidance, protein planning, resistance training, and dose adjustment.
Blood Sugar and Metabolic Improvements
Retatrutide is also being studied in type 2 diabetes. In the Phase 3 TRANSCEND-T2D-1 trial, Retatrutide lowered A1C by an average of 1.7% to 2.0% across doses at 40 weeks. Participants taking the 12 mg dose lost an average of 36.6 pounds, or 16.8% of body weight. Lilly also reported that weight loss had not plateaued by week 40.
Because Retatrutide affects multiple metabolic pathways, it may eventually become an important medication for patients with obesity, insulin resistance, prediabetes, and type 2 diabetes. However, patients taking diabetes medications must always be medically supervised because blood sugar can change significantly during treatment.
Cardiometabolic Benefits
Retatrutide studies have also shown improvements in several cardiovascular risk markers, including lipids, triglycerides, blood pressure, and other metabolic measures. These improvements are important because obesity is not only a weight issue. It is often connected to insulin resistance, inflammation, fatty liver, sleep apnea, high blood pressure, joint pain, and cardiovascular risk.
Liver Health and Fatty Liver
Retatrutide is also being studied for metabolic dysfunction-associated steatotic liver disease, often called MASLD or fatty liver disease. In a Phase 2 liver substudy, Retatrutide significantly reduced liver fat. At the two highest doses, more than 85% of participants achieved resolution of steatosis, meaning liver fat dropped below 5%.
This is one of the most exciting areas of Retatrutide research because fatty liver is closely tied to insulin resistance, abdominal weight gain, inflammation, and long-term metabolic risk.
Quality of Life
As patients lose weight, improve blood sugar, reduce joint stress, and improve metabolic function, they may experience better energy, mobility, confidence, and quality of life. In the TRIUMPH-4 study, Retatrutide was also associated with improvement in knee pain and physical function in adults with obesity or overweight and knee osteoarthritis.
When Will Retatrutide Be Available?
Retatrutide is currently in Phase 3 clinical development. It is being studied for obesity, type 2 diabetes, fatty liver disease, knee osteoarthritis, sleep apnea, chronic low back pain, and other obesity-related conditions.
There is no official FDA approval date and no official clinic launch date. The medication must complete clinical trials, be submitted to the FDA, undergo regulatory review, and receive approval before it can be prescribed in a normal clinic setting.
A reasonable expectation is that Retatrutide may reach clinics only after the remaining Phase 3 trials and FDA review are complete. Until then, any company claiming to offer real prescription Retatrutide should be approached with extreme caution.

Avoid Gray-Market Retatrutide
Do Not Buy “Research Use Only” Retatrutide Online
One of the biggest safety concerns right now is the gray market. Retatrutide is being advertised online by peptide sellers, social media sources, overseas suppliers, and “research use only” websites. These products are not FDA approved, not legally prescribed, and not held to the same accountability as legitimate medical products.
The FDA has specifically warned about unapproved GLP-1 products, including products containing semaglutide, tirzepatide, or Retatrutide that are falsely labeled “for research purposes” or “not for human consumption” while being sold directly to consumers. The FDA urges consumers not to purchase these products because they may be of unknown quality and harmful to health.
A gray-market injectable product may have problems with:
incorrect dose
wrong ingredient
contamination
poor sterility
unverified purity
improper storage
bacterial endotoxins
residual solvents or synthesis chemicals
no medical screening
no medical follow-up
no accountability if something goes wrong
Cheap online peptides can become dangerous very quickly when they are injected into the body.
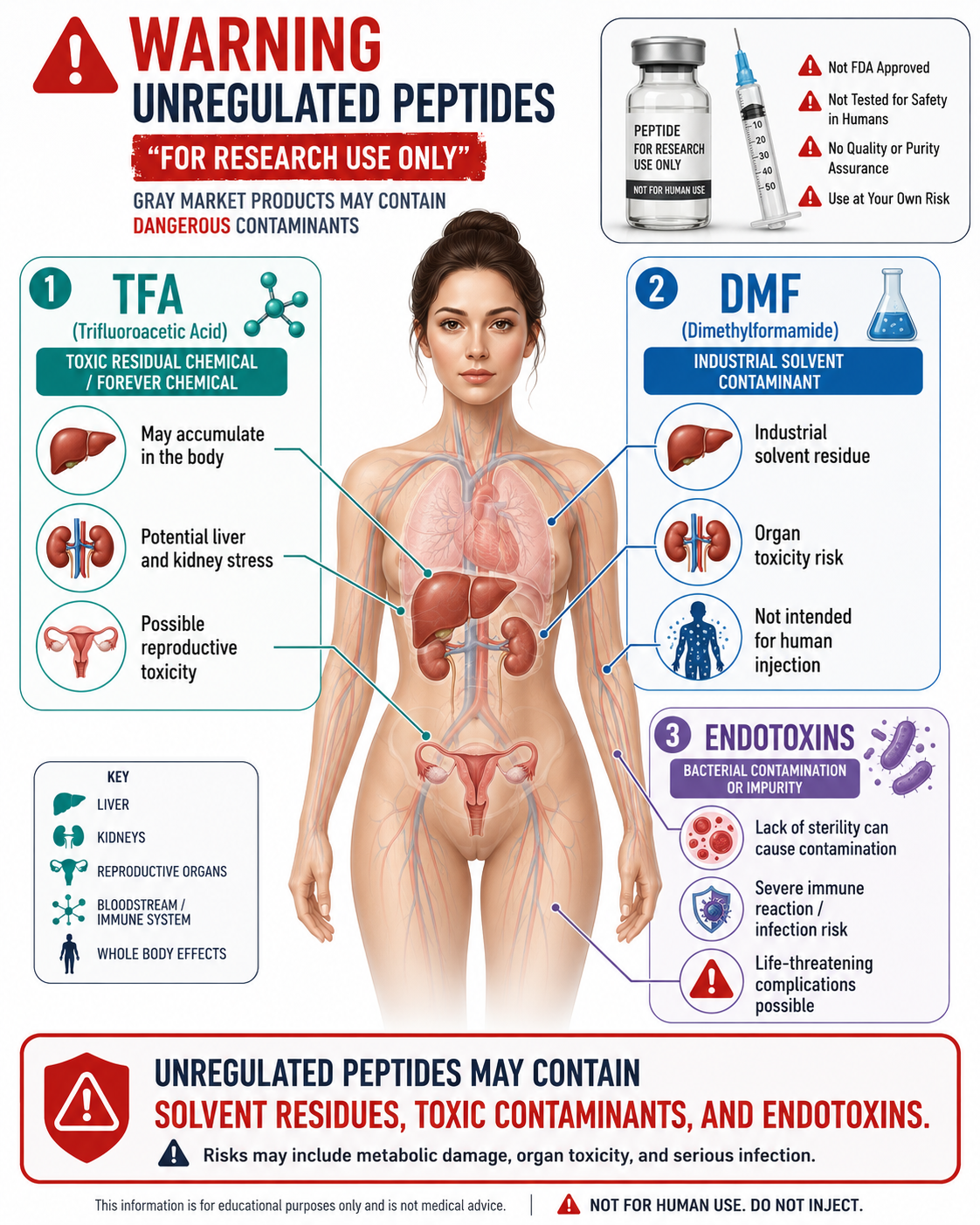
Why TFA Matters in Gray-Market Peptides
Many synthetic peptides are made using chemical processes that involve trifluoroacetic acid, commonly called TFA. TFA is often used during peptide cleavage and purification. In properly manufactured pharmaceutical products, purification and testing are used to control residues, impurities, salts, sterility, potency, and safety.
The concern with gray-market peptides is that patients usually have no reliable way to know whether residual TFA, unwanted counterions, synthesis byproducts, or other impurities have been properly removed. TFA counterions can affect the biological and physicochemical properties of peptides, which is why appropriate counterion exchange and purification matter.
This is not something a patient can verify from a label that says “99% pure.” A purity claim does not prove sterility, safety, correct dose, proper salt form, endotoxin testing, or freedom from harmful contaminants.
Why Endotoxins Matter
Endotoxins are toxic components from gram-negative bacteria. They are especially concerning in injectable products because they can trigger inflammatory and pyrogenic reactions. The FDA has detailed guidance around pyrogen and endotoxin testing for drug and biological products, including the importance of bacterial endotoxin testing standards.
Endotoxin contamination can occur through water, raw materials, equipment, handling, or poor manufacturing controls. Once present, endotoxins can be difficult to remove. This is one reason injectable medications should come from accountable, regulated, medically appropriate sources rather than unknown online sellers.
The Modern Wellness Approach to Medical Weight Loss
At Modern Wellness, we believe medical weight loss should be safe, supervised, and designed around the whole person. We do not believe patients should be guessing with gray-market peptides, “research” products, or unverified online injections.
When appropriate, Modern Wellness uses Tirzepatide from a reputable 503A compounding pharmacy source operating under the federal 503A compounding framework. This allows patient-specific prescribing through a medical provider rather than buying unknown substances online. Compounded medications are not FDA-approved drugs, but they should still come from legitimate pharmacy channels, not gray-market peptide suppliers.
Our program may also include NAD+ and lipotropic injections when appropriate. These supportive therapies are used as part of a broader wellness strategy to support energy, metabolism, cellular function, and fat-loss support. They are not magic replacements for nutrition, strength training, medical oversight, or lifestyle change. But when used thoughtfully, they can be part of a comprehensive plan.
Retatrutide Is Promising, But It Is Not Here Yet
Retatrutide may become one of the most powerful medications in the future of obesity medicine. The research is exciting, especially in weight loss, blood sugar control, fatty liver, and cardiometabolic health. But it is still investigational.
Until Retatrutide is FDA approved and available through legitimate medical channels, patients should avoid gray-market products and work with trusted medical providers using available, appropriate, and accountable treatment options.
At Modern Wellness, we help patients pursue medical weight loss in a way that is supervised, personalized, and focused on long-term health.
Why Sarcopenia Matters During GLP-1 Weight Loss
Sarcopenia means loss of muscle mass, strength, and function. It becomes more common with aging, inactivity, hormonal changes, poor protein intake, and rapid weight loss. For men and women over 40, and especially over 50, preserving muscle is one of the most important parts of a successful weight-loss plan.
GLP-1-based medications can produce excellent fat loss, but body composition still matters. In semaglutide body-composition research, total fat mass and visceral fat mass decreased, while lean mass also decreased, although the proportion of lean mass relative to total body mass improved. Tirzepatide body-composition research also shows large reductions in fat mass with some lean mass reduction, which is expected during major weight loss.
At Modern Wellness, we build the program around protecting muscle. That means adequate protein, strength training, resistance-based movement, hydration, minerals, follow-up visits, and realistic dose escalation. For many patients, the goal is not just to weigh less. The goal is to become metabolically healthier, stronger, more energetic, and more capable in daily life.
Our Weight-Loss Philosophy
Retatrutide may become an important medication in the future of obesity medicine. The early studies are promising, and the Phase 3 data so far suggest that triple-agonist therapy may offer a new level of weight-loss potential. But right now, Retatrutide is not an approved clinic medication.
Today, Modern Wellness focuses on what is available, appropriate, and medically supervised: Tirzepatide-based weight-loss care, supportive NAD+ and lipotropic options, lifestyle coaching, sarcopenia prevention, and a whole-person plan designed to help patients lose fat while protecting their strength, energy, and long-term health.
Schedule a Medical Weight Loss Consultation
If you are interested in Tirzepatide, GLP-1 weight loss support, NAD+, lipotropic injections, or a muscle-preserving weight-loss plan, Modern Wellness can help you build a program that fits your body, your goals, and your long-term health.
Modern Wellness
Medical Weight Loss • Tirzepatide Support • NAD+ • Lipotropic Injections • Muscle Preservation • Whole-Person Wellness
